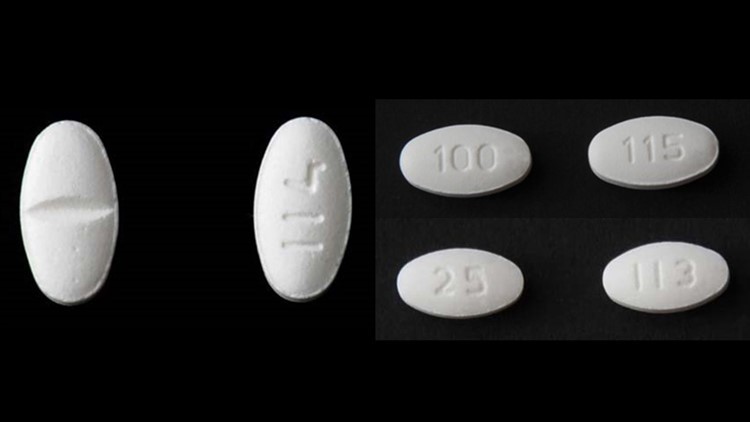
Valsartan products are used to treat high blood pressure and congestive heart failure. The way these generic drugs are being made overseas is what’s causing the recall.
Since last July eight different pharmaceutical companies have voluntarily recalled 16 different versions of medications containing Valsartan because they contain a potential carcinogen, NDMA and another impurity known as NDEA. We asked Cleveland Clinic's Chair of Cardiovascular Medicine, Dr. Steven Nissen to explain how this happened.
“They're (the drugs) being made in countries where the controls around how the drug is synthesized and what drugs are used are not as stringent as they are in the United States,” Dr. Nissen said.
He added that while the drugs are still effective, they contain small amounts of ingredients that are believed to cause cancer, but no one knows how big the cancer risk is. He adds that while the risk may be small short term, no one should be taking a drug with a known carcinogen as an ingredient.
Dr. Nissen advises patients call their pharmacy to see if the drug they’re taking is from a manufacturer where the drugs have been withdrawn. If so, ask if there’s an alternative and you may need another prescription from your doctor.
However, don’t stop taking the medication until you talk to your doctor and pharmacist.